
There is no requirement to be ISO 13485 unless specified by the customer. Is a subcontractor required to be ISO 13485 certified?
#ISO 13485 PDF FREE UPDATE#
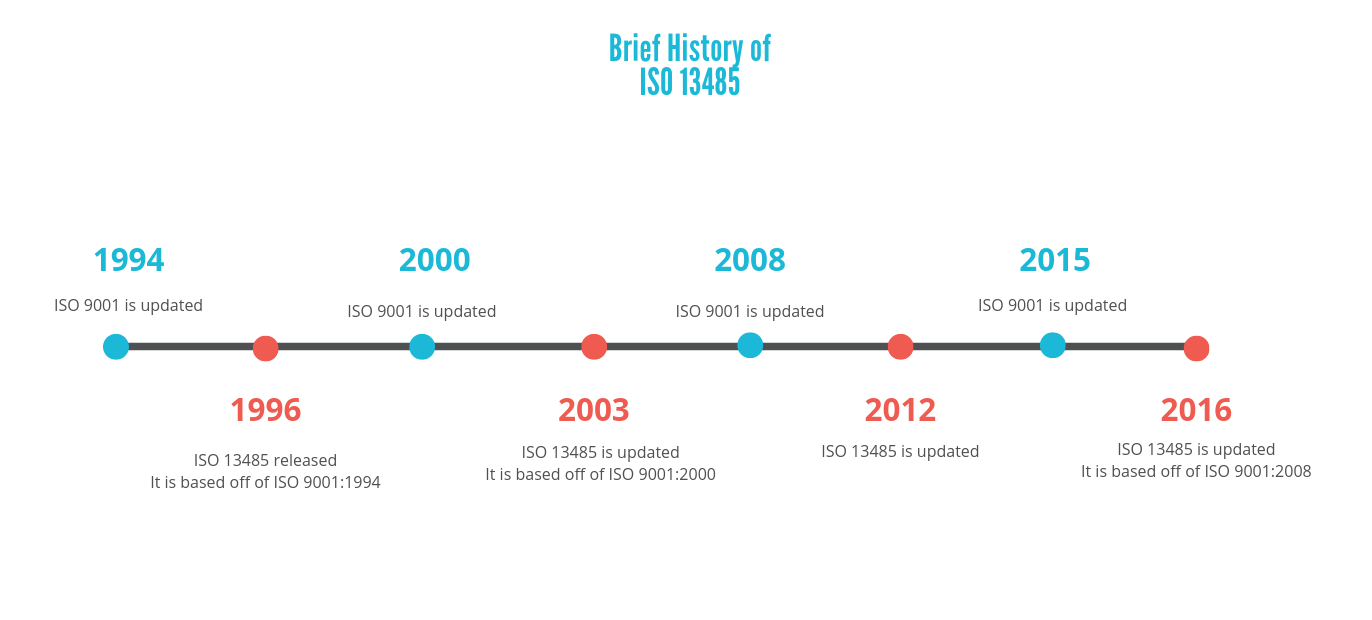
The transition period for device manufacturers across the countries that recognize the standard to update their QMS to comply with requirements under the third edition, ISO 13485:2016, is set to last until February 2019 after which the 2003 version will be withdrawn. For example, certain government bodies might issue requirements for ISO 13485 certification similarly, customers may require that their clients get certified to meet their specific needs. Do I need to certify to ISO 13485?Ĭertification isn't a requirement of ISO 13485, but it can be necessary, depending on the context. Understand and implement the quality plan, according to the applicable government requirements such as FDA 21 CFR 820, etc. This stage involves the preparation of the quality plans for implementing changes to your quality management system.
› Use Itunes To Sync Your Iphone Ipad Or Ipod With Your.
#ISO 13485 PDF FREE HOW TO#
› How To Change Password On Iphone If Forgotten.The participants will be awarded ISO 13485 Lead Auditor certificate for a trained lead auditor with successful completion of this online lead auditor training course.

This online ISO 13485 lead auditor training course will help to guide company employees of any organization as well as provides a well understanding of various activities involved in Audit Techniques, Audit Processes, and Requirements. In this ISO 13485 lead auditor training online, you will learn everything you need to know about the QMS and ISO 13485:2016 requirements as the ISO 13485 lead auditor. To fulfill all requirements of Quality Management System for medical devices, Punyam Academy’s Online ISO 13485 lead auditor training courseprovides training and certification which is very useful for users to become ‘Certified ISO 13485 lead Auditor’. ISO 13485 Lead Auditor Training Online Certification CourseĦ hours ago 1. Once this ISO 13485 auditor training is successfully completed online, participant will be qualified as a certified internal auditor with an ISO 13485 internal auditor certificate.Ĭategory: Training Courses, Online Courses, It Courses Show more The online ISO 13485 auditor trainingwill acknowledge the participants about how to perform an internal audit in such organizations. In this ISO 13485 internal auditor training online course, you will learn everything you need to know about the Quality management system and fulfill all the requirements as an ISO 13485:2016 QMS auditor. This course will guide Management as well as employees of any organization in the field of manufacturing, supply or distribution of a medical device or its components, to qualify as ‘Certified internal auditor' for Quality management system based on ISO 13485:2016 standard. For Quality management system of medical devices, the ISO 13485:2016 Auditor course provides training and certification that helps users to become Certified ISO 13485 Internal Auditor. ISO 13485 Auditor Training Online Certification Course By …ġ hours ago 1.
